

Years of experience
solutions across various industries









National service hotline
008615094320000
Circulating fluidized bed semi-dry flue gas desulfurization
Process principle
The flue gas from the boiler (kiln) furnace enters the purification tower through the air distribution device from the lower part of the purification tower after preliminary dust removal by the electrostatic precipitator. Atomized water is sprayed into the purification tower from the high-pressure reflux spray gun at the throat of the purification tower, and is mixed with the flue gas in the purification tower at a very high mass transfer rate. The small droplets and calcium oxide particles in the flue gas are mixed with the flue gas at a very high mass transfer rate. SO2 and other acidic substances in the flue gas mix and react to generate reaction products such as CaSO4 and CaSO3. A small part of these dry products are discharged from the ash discharge port at the bottom of the purification tower, and most of them are separated and collected by the bag dust collector. After the boiler flue gas is desulfurized and purified in the purification tower, it enters the bag dust collector system.
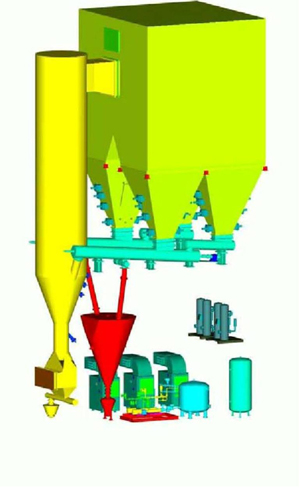
Process flow
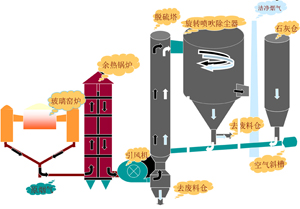
reaction process
1、 chemical process
When the atomized water passes through the high-pressure reflux (two-fluid) spray gun and is atomized in the upper part of the Venturi diffusion section of the absorption tower, and is fully contacted with the flue gas, the flue gas is cooled and humidified, and the calcium hydroxide powder particles react with H2O, SO2, and H2SO3 to form Dry powder product, the entire reaction is divided into three state reactions: gas phase, liquid phase and solid phase. The reaction steps and equations are as follows:
SO2 is absorbed by the droplets;
SO2 (gas)+H2O→H2SO3 (liquid)
The absorbed SO2 reacts with the absorbent of the solution to form calcium sulfite;
Ca(OH)2 (liquid)+H2SO3(liquid)→CaSO3(liquid)+2H2O
Ca(OH)2 (solid) +H2SO3(liquid)→CaSO3(liquid)+2H2O
Part of the CaSO3 in the solution reacts with the oxygen dissolved in the droplets and is oxidized to calcium sulfate.
CaSO3(liquid)+1/2O2(liquid)→CaSO4(liquid)
CaSO4 (liquid) has low solubility and crystallizes out
CaSO4(liquid)→CaSO4(solid)
(5) Humidify and atomize the Ca(OH)2 (solid) that has not yet reacted, and the CaO (solid) contained in CaSO3 (solid) and CaSO4 (solid).
(6) The unreacted Ca(OH)2 (solid) in the soot removed by the bag dust collector, and the Ca(OH)2 (solid) contained in CaSO3 (solid) and CaSO4 (solid) are circulated to the absorption tower Continue to react.
Principle of lime digestion: CaO(solid)+H2O (liquid)→Ca(OH)2 (solid)
2、physical process physical process
The physical process refers to the evaporation and drying of droplets and the cooling and humidification process of flue gas. The time required from the beginning of evaporation to the drying of droplets is very important to the design of the absorption tower and the desulfurization rate. Factors that affect droplet drying time include droplet size, droplet water content, and the temperature value approaching adiabatic saturation. The drying of droplets is roughly divided into two stages: the first stage, due to the small solid content in the slurry droplets, is basically free evaporation of water on the surface of the droplets, and the evaporation rate is fast and relatively constant. As the water evaporates, the solid content in the droplets increases. When significant solid matter appears on the surface of the droplets, the second stage is entered.
reaction process
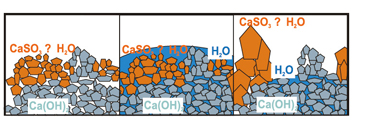
As the evaporation surface area becomes smaller, water must diffuse outward from the inside of the particles through the solid material, the drying rate decreases, the droplet temperature rises and approaches the flue gas temperature, and finally separates from the flue gas as the water evaporates to form solid particles. .
The high-rate circulation of the reaction ash in the absorption tower causes violent collisions between the circulating ash particles, destroying the solid shell of the products on the surface of the particles, and exposing the unreacted fresh particles inside to continue to participate in the reaction. Objectively, it plays the role of accelerating the reaction speed, drying speed and greatly improving the utilization rate of the absorbent. Circulating fluidized bed semi-dry desulfurization system System composition Performance guaranteed Desulfurization efficiency: ≥90% Why use circulating fluidized bed semi-dry flue gas desulfurization process? wastewater-free process Low investment cost process Ideal technology for the renovation of old factories High availability Short installation time Small footprint Low maintenance cost The final product can be sold or landfilled 
Dust emission concentration guaranteed value is below 30mg/Nm3
Ca/S:1.3-1.4
Maximum operating resistance of desulfurization and dust removal system <3200Pa
System air leakage rate ≤3%
System service life is 30 years (including filter Materials for 3 years; electromagnetic pulse valve 1 million times; high-pressure return (dual fluid) spray gun 60,000h; motors and other operating equipment are in accordance with national regulations)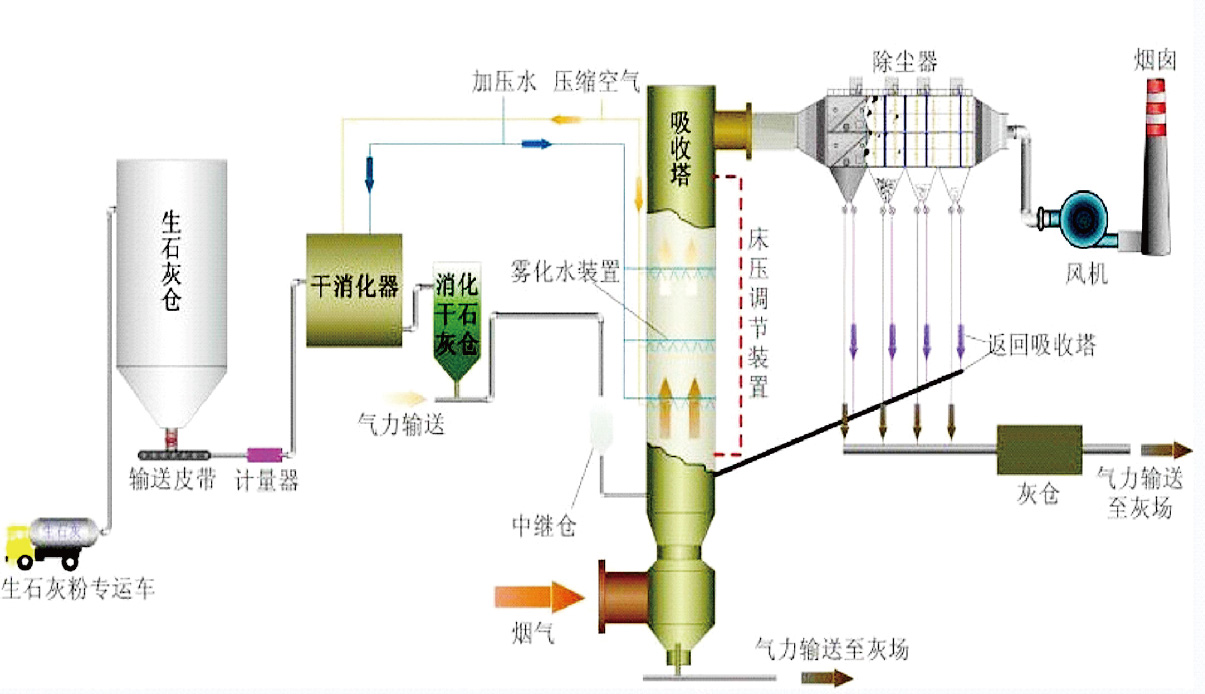
 Engineering design
Engineering design Engineering general contracting
Engineering general contracting Water operations
Water operations Environmental protection equipment
Environmental protection equipment
CopyRight © Jiangsu ZEYU Environmental Engineering Co., Ltd. Su ICP No. 14007113-1
